When navigating the world of glycols, beginners often get confused between propylene glycol (PG) and polypropylene glycol (PPG). Both are widely used in industries ranging from food and cosmetics to lubricants and pharmaceuticals, yet their properties, applications, and safety considerations differ significantly. Drawing from years of hands-on experience, this guide will break down the essential differences and help you make informed decisions.
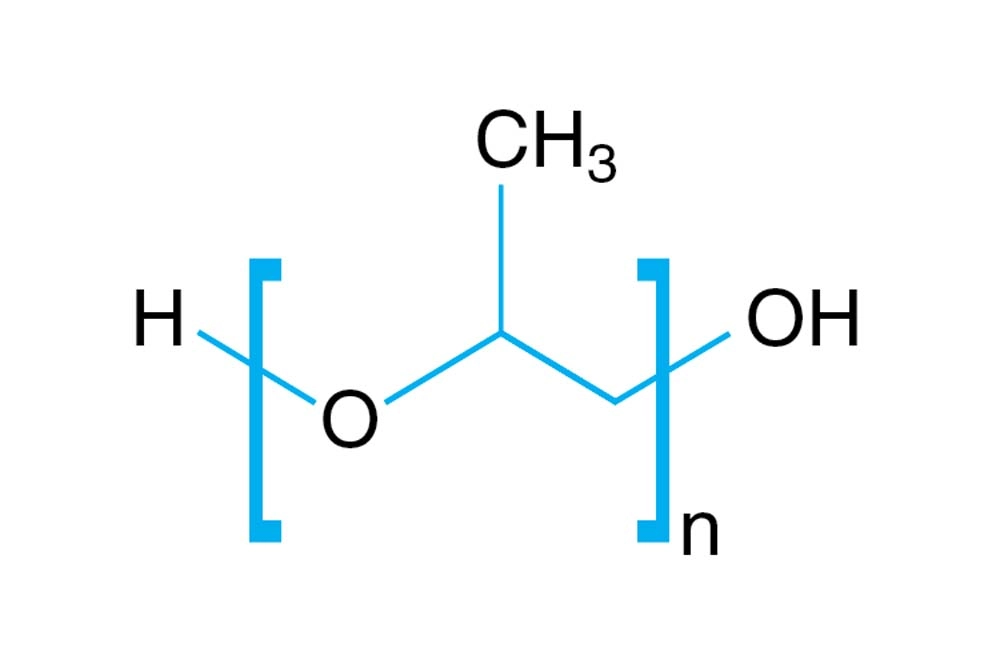
Polypropylene Glycol
Chemical structure forms the foundation for understanding PG and PPG. Propylene glycol (PG) is a small, diol molecule with the chemical formula C₃H₈O₂, containing two hydroxyl (-OH) groups attached to a three-carbon backbone. This configuration makes PG highly soluble in water and many polar solvents. Its simplicity and small size contribute to its widespread use in food, pharmaceuticals, and cosmetics.
In contrast, polypropylene glycol (PPG) is a polymer made by linking multiple propylene oxide units together, resulting in molecules of varying chain lengths and molecular weights. The repeating units create a flexible backbone, which significantly increases viscosity and reduces water solubility compared to PG. PPG is not typically used in food or personal care products, but its polymeric nature makes it ideal for lubricants, antifreeze formulations, and industrial applications.
Understanding the molecular distinction helps explain why PG excels in applications requiring water miscibility and low viscosity, whereas PPG is favored when hydrophobicity and high molecular weight are beneficial.
Physical characteristics further separate PG from PPG in practical applications. Propylene glycol is a low-viscosity liquid, with a boiling point around 188°C and excellent water solubility. These traits allow it to function effectively as a solvent, humectant, and carrier fluid in formulations where rapid dispersion is needed.
On the other hand, polypropylene glycol comes in various viscosities depending on its molecular weight. Low molecular weight PPG may be somewhat water-soluble, but high molecular weight variants are essentially hydrophobic oils. The boiling point of PPG increases with molecular weight, often exceeding 250°C, making it more suitable for heat-resistant industrial applications.
Recognizing these differences ensures the correct glycol is selected for processes requiring thermal stability, solvent compatibility, or controlled viscosity.
Applications often dictate whether PG or PPG is the better choice. Propylene glycol shines in industries where safety and biocompatibility are critical. It is widely used in food additives, flavor carriers, pharmaceuticals, e-cigarettes, and personal care products. Its water solubility and low toxicity make it a reliable ingredient in products ingested or applied to the human body.
Polypropylene glycol, due to its polymeric structure, finds favor in lubricants, hydraulic fluids, polyurethane production, and antifreeze formulations. PPG is often selected when high viscosity, lubricity, or hydrophobic behavior is desired, such as in industrial machinery and polymeric materials.
For manufacturers, the choice is often guided by specific performance requirements, regulatory considerations, and desired product characteristics.
Safety is paramount when handling glycols. Propylene glycol is generally recognized as safe (GRAS) for food and pharmaceutical use. Its toxicity is low, though excessive ingestion can cause minor gastrointestinal discomfort. Dermal exposure is typically safe, which contributes to its widespread use in cosmetics and topical formulations.
Polypropylene glycol, while non-toxic in small industrial doses, is not intended for oral or cosmetic applications. Handling PPG requires standard industrial safety measures, including ventilation, gloves, and eye protection, particularly for high-molecular-weight grades that can be viscous and slippery. Awareness of these safety profiles helps manufacturers comply with OSHA, FDA, and REACH regulations.
Selecting the right glycol involves weighing several factors: application type, solubility requirements, viscosity needs, safety considerations, and cost. For food, pharmaceutical, or cosmetic formulations, PG is almost always the preferred choice due to its biocompatibility and water miscibility. For industrial applications involving lubricants, heat transfer fluids, or polymer production, PPG often provides superior performance.
Additional considerations include chain length and molecular weight, which directly influence viscosity and hydrophobicity in PPG, and formulation compatibility, which determines how well PG or PPG mixes with other ingredients. Manufacturers must balance performance, regulatory compliance, and economic feasibility when choosing between these glycols.